Aurora project overview
The MYC family of cellular oncogenes encodes three highly related nuclear phosphoproteins (c-Myc, N-Myc, and L-Myc) that are believed to function as sequence-specific transcription factors capable of regulating genes important in cellular growth and differentiation. [1] Due to this involvement in central cellular processes they play a key role in development and maintenance of a multitude of human cancer diseases. Cellular MYC concentration is tightly regulated at the level of protein stability through canonical upstream kinase signaling pathways, including phosphatidylinositol 3-kinase (PI3K)/mammalian target of rapamycin (mTOR), cyclin-dependent kinase 2 (CDK2), and mitogen-activated protein kinase (MAPK). These kinases direct sequential phosphorylation and dephosphorylation of conserved residues in MYC proteins, which target them for ubiquitination followed by proteasomal degradation. [2] Gustafson et al. showed in 2014 that Aurora Kinase A protects N-MYC against degradation by forming a protein-protein-interaction. This stabilization was interrupted by conformation disrupting inhibitors not acting like classical type I Kinase inhibitors. Additionally, Dauch et al. produced similar results for c-MYC, after siRNA-screens detected an increased Aurora Kinase A levels in a MYC-dependent hepatocellular carcinoma cell line, confirming that appropriate inhibitors show the desired biological effect. In-silico-modelling showed a possible explanation for the observed biological effects of conformation disrupting inhibitors. Aurora Kinase A seems to mask phosphor-residues that target MYC for ubiquitination, subsequent preventing formation of the MYC/Aurora complex should result in proteasomal degradation of MYC (fig. 1). [3]
Our studies focus on design and synthesis of decent Aurora Kinase A inhibitors providing structural features that are able to prevent complex formation to ultimately achieve the degradation of MYC - a target, that is considered undruggable.
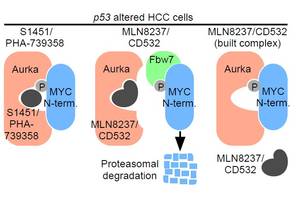
Figure 1: Schematic mode of action; left: Type-I-Inhibitors enable the interaction between phosphorylated c‑MYC; middle: conformation-disrupting-Inhibitors suppress formation of the protein-protein-complex; right: already built complexes are not affected. [3]
[1] B. Mukherjee, S. D. Morgenbesser, R. A. Depinho, Gene Dev 1992, 6, 1480-1492.
[2] W. C. Gustafson, J. G. Meyerowitz, E. A. Nekritz, J. Chen, C. Benes, E. Charron, E. F. Simonds, R. Seeger, K. K. Matthay, N. T. Hertz, M. Eilers, K. M. Shokat, W. A. Weiss, Cancer Cell 2014, 26, 414-427.
[3] D. Dauch, R. Rudalska, G. Cossa, J. C. Nault, T. W. Kang, T. Wuestefeld, A. Hohmeyer, S. Imbeaud, T. Yevsa, L. Hoenicke, T. Pantsar, P. Bozko, N. P. Malek, T. Longerich, S. Laufer, A. Poso, J. Zucman-Rossi, M. Eilers, L. Zender, Nat Med 2016, 22, 744-753.
