Research Interests of the Bettinger Group
Boron Containing Reactive Intermediates
The study of reactive intermediates lies at the heart of mechanistic chemistry. Our research focuses on neutral reactive boron containing compounds including borylenes, borylnitrenes, aminoborylenes, and azaborines. These correspond to the well-known carbenes, vinylidenes, and benzynes, respectively.
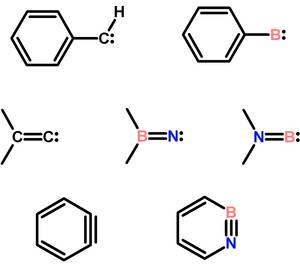
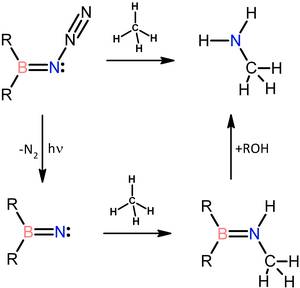
Various pathways are employed towards these intermediates and their properties are being studied by low temperature spectroscopy and evaluated in synthetic transformations. Borylnitrenes, for example, can be generated photochemically from borylazides and be used for room temperature C–H transformation of unactivated hydrocarbons, including even the unreactive methane molecule.
Representative Publications:
Noble Gas Atoms as Electron Donors: Is the Stabilization of Strongly Electrophilic Borylnitrenes Feasible?
Bettinger, H. F.; Bornemann, H. Z. Anorg. Allg. Chem. 2011, 637, 2169-2174.
Synthesis and Structural Characterization of 9-Azido-9-Borafluorene: Monomer and Cyclotrimer of a Borole Azide.
Biswas, S.; Oppel, I. M.; Bettinger, H. F.; Inorg. Chem. 2010, 49, 4499-4506.
Splitting of Dihydrogen Employing the Nitrogen Center of a Borylnitrene.
Bettinger, H. F.; Filthaus, M.; Neuhaus, P. Chem. Commun. 2009, 2186-2188.
Metal-Free Conversion of Methane and Cycloalkanes to Amines and Amides Employing a Borylnitrene.
Bettinger, H. F.; Filthaus, M.; Bornemann, H.; Oppel, I. M. Angew. Chem. Int. Ed. 2008, 47, 4744-4747.
BN-Analogues of Vinylidene Transition Metal Complexes: The Borylnitrene Isomer.
Bettinger, H. F. Inorg. Chem. 2007, 46, 5188-5195.
Donor Stabilized Borylnitrene: A Highly Reactive BN Analogue of Vinylidene.
Bettinger, H. F.; Bornemann, H. J. Am. Chem. Soc. 2006, 128, 11128-11134.
Phenylborylene: Direct Spectroscopic Investigation in Inert Gas Matrices.
Bettinger, H. F. J. Am. Chem. Soc. 2006, 128, 2534-2535.
Acenes
Acenes, composed of linearly annulated benzenes, experience significant interest as organic semiconducting materials. Unfortunately, their reactivity increases very quickly with length mainly due to oxidative decomposition. This is where our interests start: we focus on the synthesis, characterization, and investigation of the chemical properties of acenes that are too unstable under ordinary laboratory conditions.
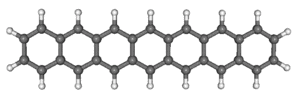
Representative Publications:
The Influence of Terminal Push-Pull Substitution on the Electronic Structure and Optical Properties of Pentacenes.
Tönshoff, C.; Bettinger, H. F. Chem. Eur. J. 2012, 18, 1789-1799.
Photogeneration of Octacene and Nonacene.
Tönshoff, C.; Bettinger, H. F. Angew. Chem. Int. Ed. 2010, 49, 4125-4128.
Electronic Structure of Higher Acenes and Polyacene: The Perspective Developed by Theoretical Analyses.
Bettinger, H. F. Pure Appl. Chem. 2010, 82, 905-915.
Synthesis, Stability, and Photochemistry of Pentacene, Hexacene, and Heptacene: A Matrix Isolation Study.
Mondal, R.; Tönshoff, C.; Khon, D.; Neckers, D. C.; Bettinger, H. F. J. Am. Chem. Soc. 2009, 131, 14281-14289.
The Shapiro Reaction of Barrelene Derivatives: The Influence of Annelation on Acene Formation.
Bettinger, H. F.; Mondal, R.; Tönshoff, C. Org. Biomol. Chem. 2008, 6, 3000-3004.
Stable Photoinduced Charge Separation in Heptacene.
Bettinger, H. F.; Mondal, R.; Neckers, D. C. Chem. Commun. 2007, 2509-2511.
BN Containing Polycyclic Aromatic Hydrocarbons
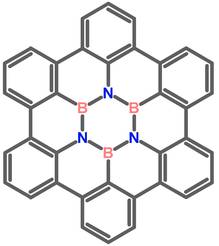
We are also interested in novel compounds that include boron-nitrogen units within the carbon framework of polycyclic aromatics. Such BN-doped derivatives are currently receiving attention as promising novel organic-inorganic materials for electronic applications. Our research aims at the synthesis of such new materials that could be used in transitors, photovoltaics, or organic light emitting diodes.
Representative Publication:
BN-doped Graphene Molecules: Hexa-peri-hexabenzocoronene with a B3N3 Borazine Core and Its Attempted Synthesis by a Scholl Reaction.
Tönshoff, C.; Müller, M.; Kar, T.; Latteyer, F.; Chassé, T.; Eichele, K.; Bettinger, H. F. Chem. Phys. Chem. 2012, accepted for publication.
