PII-signal tranduction systems in bacteria
PII signal transduction proteins are one of the most widely distributed and highly conserved signalling proteins in nature. They play key roles in global nitrogen control by acting as a signal node that perceives signals from central metabolism and transmits a coherent response to various targets to balance C- and N- metabolism. PII proteins are homotrimers with complex inter-subunit communication between the binding site of the effector metabolites ATP, ADP and 2-OG (2-oxoglutarate).
The Forchhammer lab is exploring the role, structure and function relation of cyanobacterial PII proteins.
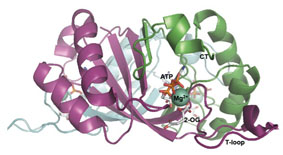
PII protein with bound ATP-Mg2+-2OG
Recently we could identify the site of 2-OG binding, located in the vicinity of the ATP-binding site. In addition to responding to nitrogen levels, binding of ATP and ADP, which compete for the same binding site, allows PII also to sense changing adenylate energycharge and control its target proteins accordingly.In Synechococcus elongatus, PII binds a small protein termed PipX, which is a co-activator of the transcription factor NtcA, and regulates the key enzyme of the cyclic ornitine pathway, N-acetyl-L-glutamate kinase.The interaction of PII with NAGK plays an important role in the cellular metabolism. We could show that PII and NAGK from bacteria (S. elongatus) and plants (Arabidopsis thaliana) can functionally complement each other, demonstrating a strong conservation of this regulatory mechanism through 1.2 billion years of separate evolution.
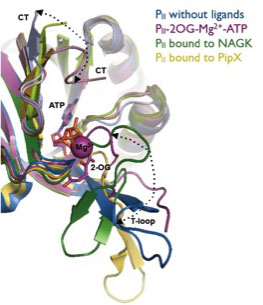
Conformational states of PII
PII proteins also integrate metabolic signals by being covalently modified. In unicellular cyanobacteria of the genera Synechococcus and Synechocystis, PII responds to changing carbon and nitrogen availability by phosphorylation/dephosphorylation at a seryl-residue located at a surface-exposed loop structure. The PII phosphatase, which was identified in Synechocystis sp. PCC 6803, is a member of PPM/PP2C family of protein phosphatases, widely distributed in prokaryotes and eukaryotes. Mutational studies and biochemical analysis revealed insight into the molecular mechanisms of substrate recognition by this enzyme.
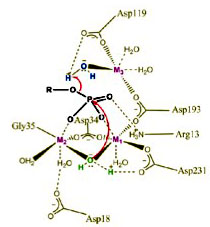
Catalytic mechanism of the PII phosphatase
A FRET sensor was developed which gives relativ information on the interaction of PII with NAGK and could be used as a sensor for 2-Oxo-glutarate.
For recent publications see list of publications
